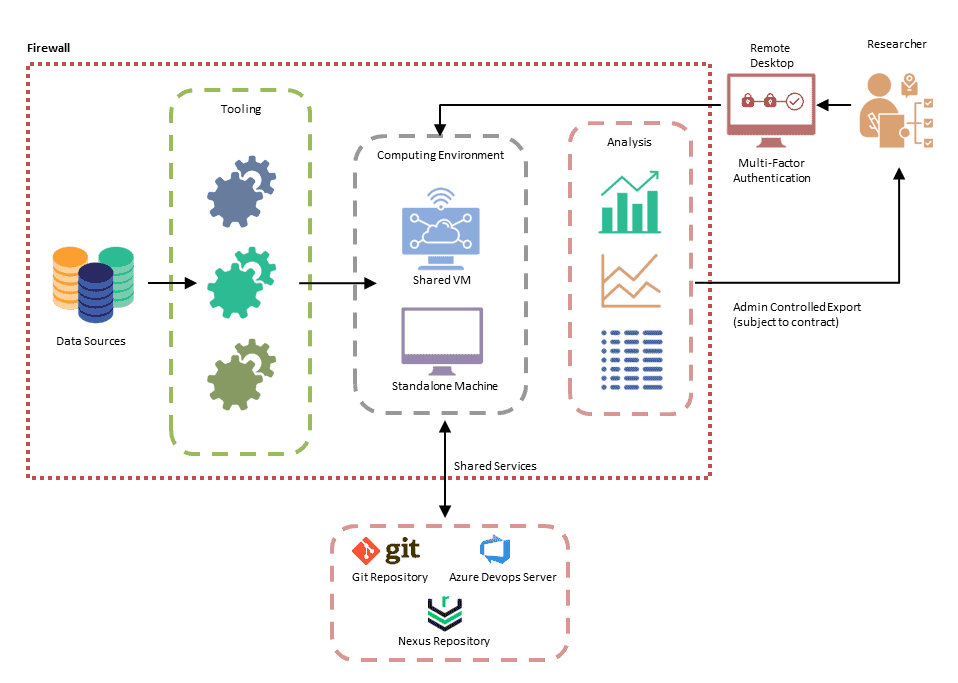
Data Access
Data access is available via our Trusted Research Environments (created to meet your exact requirements) or your own, provided it is secure.
Please contact us for more information.
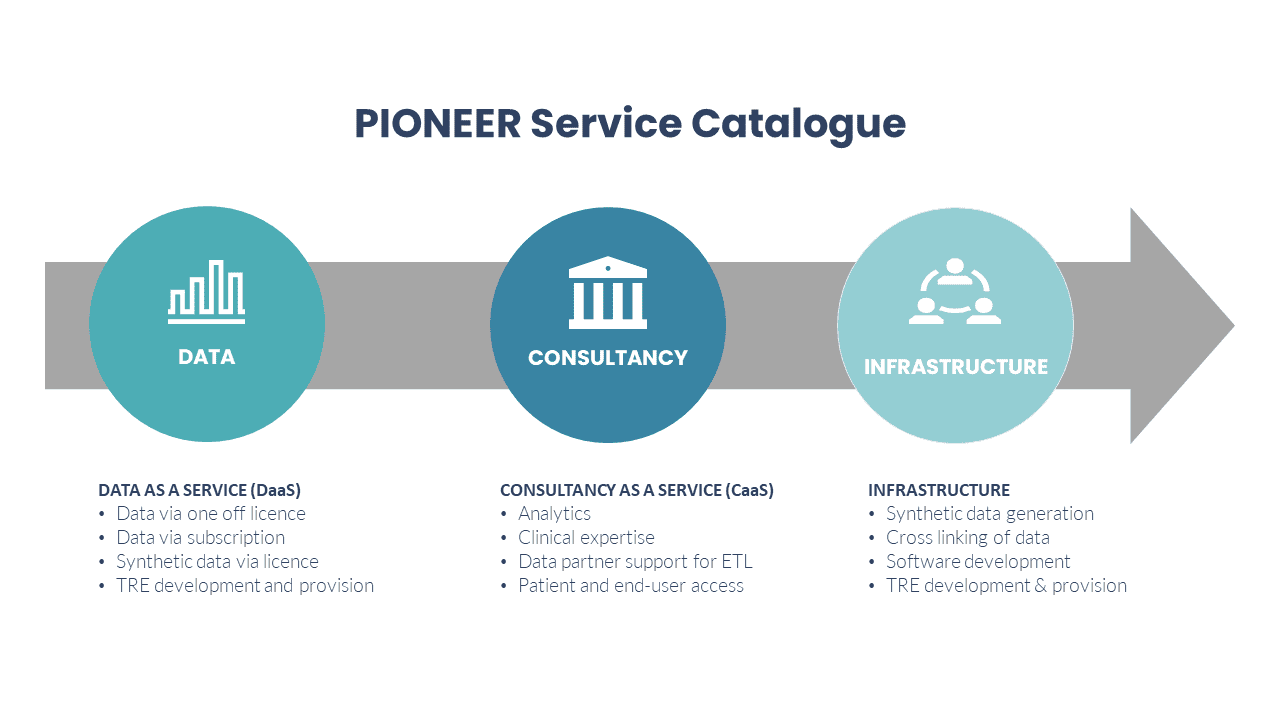
Data and Infrastructure Consultancy
These services are provided by our expert team of analysts, technologists and clinical staff.
- Cross linking of data across healthcare providers including harmonising different data systems.
- High quality synthetic data generation
- Imaging data
- Artificial Intelligence (AI) algorithm expertise
- Software development
- Data insight & problem solving
- Regulatory advice
- Patient and purchaser engagement
- Trusted/Secure Research Environment development and provision, to meet your data analysis needs for any project.
PIONEER can assist with:
- Head-to-head comparisons of investigations, products, procedures or pathways
- Assessing side effects and drug interactions
- Real world survival/efficacy
- Pre-market/Post-market
- Population size and make up.
- Representative clinical trial data
- Uptake criteria



